In the world of orthopedics and sports, we often unintentionally overlook the neurological contribution to rehabilitation. Although most practitioners don’t completely neglect the brain and are incorporating some sort of neuromuscular training into their rehab programs, they often struggle to articulate the science behind their methods. Here we’re going to dive into the ‘why’ behind using neuromuscular training in rehabilitation and how mTrigger visual sEMG biofeedback can aid in that process.
First, let’s break down some important definitions:
- Motor control is the process of initiating, directing, and grading purposeful voluntary movement.
- Motor learning is an individual’s ability to acquire and refine motor skills with relatively permanent change through repeated practice or experiences.(1)
- Neuromuscular training is defined as training that incorporates functional and sport specific movements, such as strength and conditioning activities, resistance training, dynamic stability, balance, core strength, plyometrics, and agility – with the overall goal of enhancing health and skills related to fitness and injury prevention.(2)
- Somatosensation is the perception of sensory stimuli that involves touch, temperature, body position, and pain.
These concepts play an integral role in the rehabilitation and training progressions after an injury. Although we will mostly focus on the science behind ACL motor learning in the paragraphs to follow, these overarching concepts can be applied across the board for injuries presenting with neuromuscular deficits.
70% of ACL injuries are non-contact injuries; they primarily occur when the knee falls into a high injury risk position due to motor coordination errors resulting from nervous system failure.(3,4) Movement control is accomplished through the planning and execution of complex neural processes, detected by the integration of sensory input from the body and external environment.(4) In lay terms, our brain interprets signals from our body and the environment to then produce the appropriate movement. Athletes are required to perform tasks in response to frequently-changing temporal (relating to timing) and spatial (relating to space) demands.(4)
For example:
- A soccer player predicting when (temporal) and where (spatial) to jump to head a moving soccer ball
- A football player trying to dodge a tackle while still catching the ball
- A baseball player trying to catch the ball then tag the runner
- A gymnast flipping and twisting in the air before finding the landing mat
Tasks requiring both spatial and temporal dimensions increase brain activity and connectivity within the sensory and cerebellar regions.(4) Conversely, spatial tasks alone merely activate the cerebellar regions of the brain and are much easier to accomplish.(4) Given this, compromised knee positions are more likely to occur under spatially and temporally demanding situations.(4) For example, situations that require attending to an external focus with highly demanding visual stimuli, variable surfaces, complex movement patterns, rapid changes in movement, and/or unpredictable player/environment interactions would be considered spatially and temporally demanding.(5) Hence, the integration of motor learning and neuromuscular re-training becomes critical following an injury.
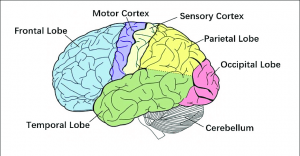
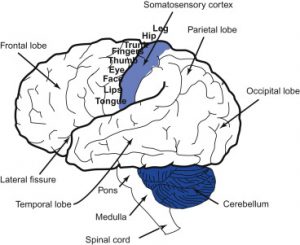
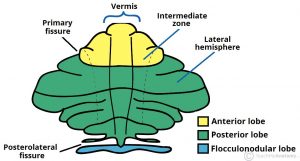
Diekfuss and his team investigated central nervous system (CNS) differences between athletes who have sustained an ACL injury and those who have not.(4) They found there was a decrease in functional connectivity between the left primary sensory cortex and the right posterior lobe of the cerebellum prior to knee injury.(4) The left primary sensory cortex plays a large role in processing sensory signals and guiding the motor actions that are to follow; the cerebellum controls postural stability.(4) The decreased connectivity between these brain areas seen in the ACL rupture group makes sense given the deficits mentioned above.(4)
In another study, ACL trauma resulted in changes in the interaction between vision and somatosensation in the nervous system.(5) The visual system is fundamental for providing coordination, regulation, and control of movement with increasingly difficult, complex, and variable tasks.(5) Combined with vision, the somatosensory system provides input to the central nervous system to control movement.(5) In trauma cases, injury disrupts normal neural activation and communication, heightening re-injury risk.
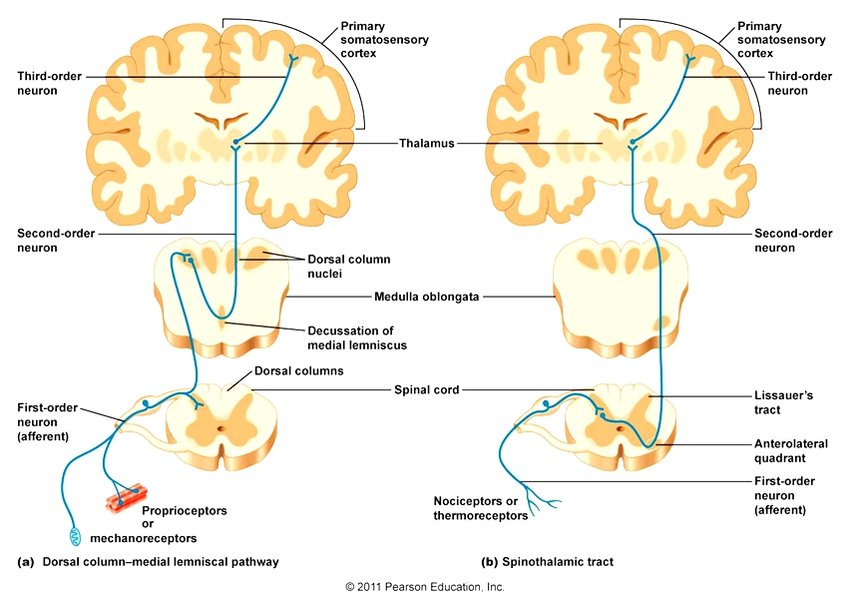
If neuromuscular deficits exist before an injury, they certainly are not going to get better after an injury; they must be directly and purposefully addressed. After an ACL injury, there is a loss of mechanoreceptors within the knee joint; this leads to a disruption in the afferent pathways (ascending information to the brain), causing motor compensations, and nervous system changes.(4,5) The loss of primary somatosensory afferent information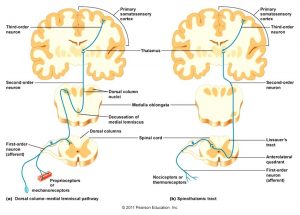
As we can see, it is important to target the nervous system changes associated with faulty movement patterns. We can achieve this by integrating neurocognitive demands and neuromuscular rehabilitation progressions to improve the transfer of sensorimotor adaptations achieved in rehab to everyday life.(5) But, enough of the nerdy science- in Part 2 of this post we will look at what this means clinically and how to use it practically.
Check out this Case Study on Neuromuscular Deficits
|
Sign Up for Our E-mail List
|
References
- Gokeler A, Neuhaus D, Benjaminse · Anne, Grooms DR, Baumeister J. Principles of Motor Learning to Support Neuroplasticity After ACL Injury: Implications for Optimizing Performance and Reducing Risk of Second ACL Injury Key Points. Sport Med. 2019;49:853-865. doi:10.1007/s40279-019-01058-0
- Granacher U, Puta C, Gabriel HHW, Behm DG, Arampatzis A. Editorial: Neuromuscular training and adaptations in youth athletes. Front Physiol. 2018;9(SEP):1264. doi:10.3389/FPHYS.2018.01264/BIBTEX
- Letafatkar A, Rajabi R, Tekamejani EE, Minoonejad H. Effects of perturbation training on knee flexion angle and quadriceps to hamstring cocontraction of female athletes with quadriceps dominance deficit: Pre-post intervention study. Knee. 2015;22(3):230-236. doi:10.1016/j.knee.2015.02.001
- Diekfuss JA, Grooms DR, Yuan W, et al. Does Brain Functional Connectivity Contribute to Musculoskeletal Injury? A Preliminary Prospective Analysis of a Neural Biomarker of ACL Injury Risk HHS Public Access. J Sci Med Sport. 2019;22(2):169-174. doi:10.1016/j.jsams.2018.07.004
- Grooms D, Appelbaum G, Onate J. Neuroplasticity following anterior cruciate ligament injury: A framework for visual-motor training approaches in rehabilitation. J Orthop Sports Phys Ther. 2015;45(5):381-393. doi:10.2519/JOSPT.2015.5549
- Miko SC, Simon JE, Monfort SM, Yom JP, Ulloa S, Grooms DR. Postural stability during visual-based cognitive and motor dual-tasks after ACLR. J Sci Med Sport. 2021;24(2):146-151. doi:10.1016/J.JSAMS.2020.07.008
- Benjaminse A, Otten B, Gokeler A, Diercks RL, Koen ·, Lemmink APM. Motor learning strategies in basketball players and its implications for ACL injury prevention: a randomized controlled trial. Knee Surgery, Sport Traumatol Arthrosc. 2017;25:2365-2376. doi:10.1007/s00167-015-3727-0
Media Sources:
https://www.sciencedirect.com/topics/medicine-and-dentistry/primary-somatosensory-cortex
https://teachmeanatomy.info/neuroanatomy/structures/cerebellum/








Leave A Comment